Chemistry, 18.03.2021 01:20, jackfrost5
Hydrogen sulfide gas reacts with oxygen to produce sulfur dioxide gas and water according to the balance equation:
2H2S + 3O2 → 2SO2 + 2H2O.
How many moles of oxygen gas are required to react with 5.6 moles of hydrogen sulfide?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 12:00, angieplasencia8
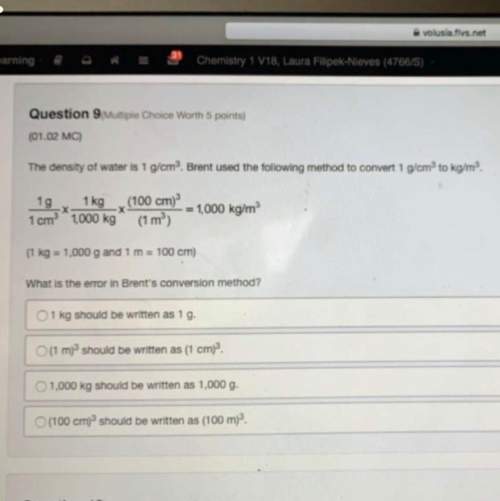
Which element has the largest atomic radius? a. asb. nc. pd. sb
Answers: 2

Chemistry, 23.06.2019 17:00, bradenjesmt1028
An unknown substance was put in a container. the substance filled up the container from bottom up, taking half of the space in the container. what is most likely the state of matter of the substance? gas liquid plasma solid
Answers: 3

Chemistry, 23.06.2019 17:30, evelynnn511
What amount in moles does 242 l of carbon dioxide occupy at 1.32 atm and 20 degrees c?
Answers: 2
Do you know the correct answer?
Hydrogen sulfide gas reacts with oxygen to produce sulfur dioxide gas and water according to the bal...
Questions in other subjects:





SAT, 28.12.2021 05:30

SAT, 28.12.2021 05:30