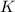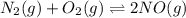Chemistry, 18.03.2021 01:10, alexandrecarmene
The equilibrium-constant expression is used to describe the concentration of reactants and products for a reaction in dynamic equilibrium. For ideal gases and ideal solutions in homogeneous equilibria, where all reactants and products are in the same phase, the extent to which a particular chemical reaction proceeds to products is given by the equilibrium equation.
aA+bB⇌cC+dD , K=[C]c[D]d[A]a[B]b
where K is the equilibrium constant and the right-hand side of the equation is known as the equilibrium-constant expression.
The concentration of each product raised to its coefficient is divided by the concentration of each reagent raised to its coefficient according the the balanced chemical equation. Therefore, the higher the concentration of products, the larger the value of K will be.
Required:
Identify the proper form of the equilibrium-constant expression for the equation.
N2(g)+O2(g)⇌2NO(g)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:10, bri9263
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 23.06.2019 00:30, ejuarez2020
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 08:40, mathisaqeosmw
Calculate the number of grams of sodium in 3.00 g of each sodium-containing food additive.
Answers: 3

Do you know the correct answer?
The equilibrium-constant expression is used to describe the concentration of reactants and products...
Questions in other subjects:






Social Studies, 06.01.2020 19:31



Social Studies, 06.01.2020 19:31

Computers and Technology, 06.01.2020 19:31


![K=\frac{[NO]^2}{[N_2]^1[O_2]^1}](/tpl/images/1195/8832/df6dc.png)