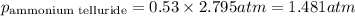Chemistry, 17.03.2021 23:40, brianna1096
A tank consists of a gaseous mixture of 3.83 g of triselenium diiodide, 2.62 g of ammonium telluride, and 1.82 g of iridium (i) sulfate. What is the partial pressure in atm of ammonium telluride if the total pressure in the tank is 2.795 atm?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 09:00, bibhu42kumarp7o4ss
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 09:10, chloeholt123
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2
Do you know the correct answer?
A tank consists of a gaseous mixture of 3.83 g of triselenium diiodide, 2.62 g of ammonium telluride...
Questions in other subjects:




Biology, 15.11.2019 09:31


Chemistry, 15.11.2019 09:31




Mathematics, 15.11.2019 09:31


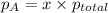
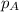 = partial pressure of A
= partial pressure of A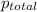 = total pressure
= total pressure 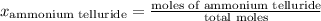 ,
,