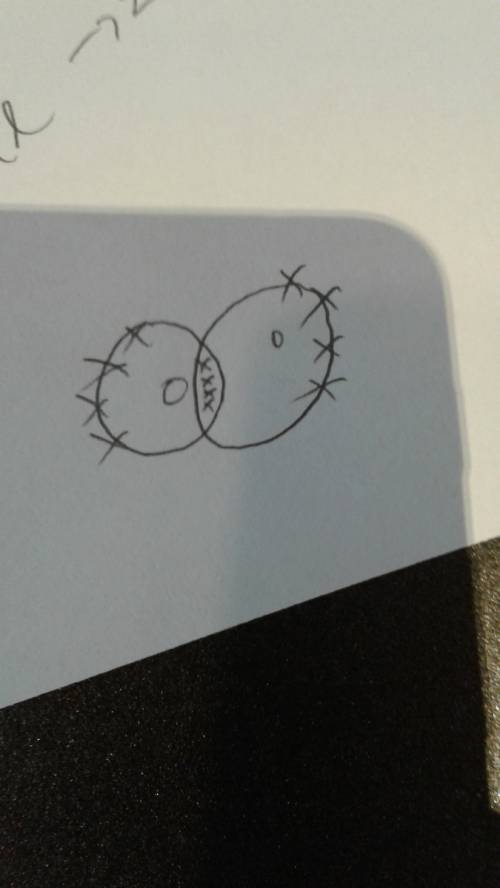
An oxygen atom has six electrons in its outermost energy level.
explain why two oxygen at...

Chemistry, 29.09.2019 18:30, BreBreDoeCCx
An oxygen atom has six electrons in its outermost energy level.
explain why two oxygen atoms must share four electrons when they form a covalent bond?
answer in complete sentences and thoroughly. 5-7

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3


Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01

Mathematics, 16.09.2020 03:01