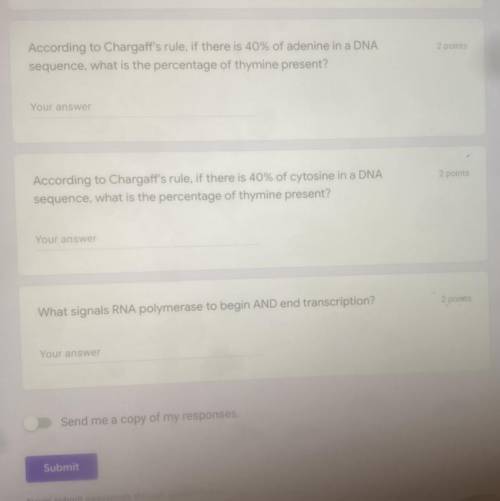
HELPP PLZZZ THANKSSS
...

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 00:30, Keemdadream13
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 06:00, wirchakethan23
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 28.06.2019 22:30



History, 28.06.2019 22:30

English, 28.06.2019 22:30

English, 28.06.2019 22:30

History, 28.06.2019 22:30


Mathematics, 28.06.2019 22:30

History, 28.06.2019 22:30