
Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:40, jaueuxsn
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 09:00, dante766
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1
Do you know the correct answer?
Write the electron configuration for . (Express your answer as a series of orbitals, in order of inc...
Questions in other subjects:

Mathematics, 01.03.2021 04:40



Business, 01.03.2021 04:40



Biology, 01.03.2021 04:40



Advanced Placement (AP), 01.03.2021 04:40


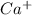 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 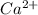 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 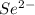 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.) Write the electron configuration for 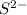 . (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)
. (Express your answer as a series of orbitals, in order of increasing orbital energy. For example, the electron configuration of Li would be entered as 1s22s1 or [He]2s1.)![Ca^+:19:[Ar]4s^1](/tpl/images/1191/0394/b80e3.png)
![Ca^{2+}:18:[Ar]](/tpl/images/1191/0394/ad5ef.png)
![Se^{2-}:32:[Ar]3d^{10}4s^24p^2](/tpl/images/1191/0394/c1b03.png)
![S^{2-}:18:[Ar]](/tpl/images/1191/0394/b3fdb.png)
![Ca:20:[Ar]4s^2](/tpl/images/1191/0394/46427.png)
![Se:34:[Ar]3d^{10}4s^24p^4](/tpl/images/1191/0394/0cf30.png)
![Se^{2-}:36:[Ar]3d^{10}4s^24p^6](/tpl/images/1191/0394/999f5.png) or
or ![Se^{2-}:36:[Kr]](/tpl/images/1191/0394/523d5.png)
![S:16:[Ne]3s^23p^4](/tpl/images/1191/0394/70407.png)
![S^{2-}:18:[Ne]3s^23p^6](/tpl/images/1191/0394/4203d.png) or
or 



