
Chemistry, 11.03.2021 04:40, savageyvens
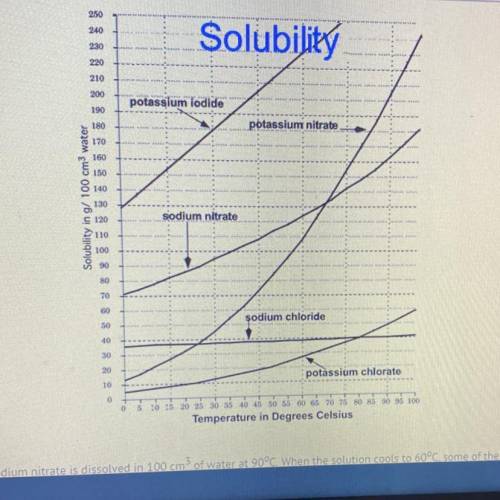
160 grams of sodium nitrate is dissolved in 100 cm of water at 90°C. When the solution cools to 60°C, some of the salt settles out
in the bottom of the beaker. The salt settles out because the solution is now
A)
saturated
B)
unsaturated
oversaturated
D)
supersaturated

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:40, ohgeezy
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1



Chemistry, 23.06.2019 01:30, nikonee
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
Do you know the correct answer?
160 grams of sodium nitrate is dissolved in 100 cm of water at 90°C. When the solution cools to 60°C...
Questions in other subjects:



Mathematics, 16.02.2021 01:30


Mathematics, 16.02.2021 01:30

Mathematics, 16.02.2021 01:30










