
Chemistry, 11.03.2021 04:30, Dracopaul03
HELP!!
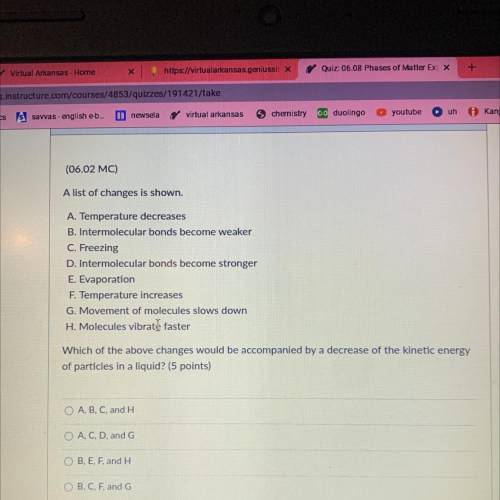
A list of changes is shown.
A. Temperature decreases
B. intermolecular bonds become weaker
C. Freezing
D. Intermolecular bonds become stronger
E. Evaporation
F. Temperature Increases
G. Movement of molecules slows down
H. Molecules vibrate faster
Which of the above changes would be accompanied by a decrease of kinetic energy of particles in a liquid?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 02:40, towelmearowel
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 03:30, antoinetteee03
Name atleast 3 type of energy associated with the microwave
Answers: 1
Do you know the correct answer?
HELP!!
A list of changes is shown.
A. Temperature decreases
B. intermolecular bonds bec...
A. Temperature decreases
B. intermolecular bonds bec...
Questions in other subjects:















