
Chemistry, 10.03.2021 01:40, mshields1994
Dihydrogen sulfide reacts with oxygen gas to form sulfur dioxide and water. How many moles of oxygen gas will react when 0.6469 moles of water are formed?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, jbarbie3
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1

Chemistry, 22.06.2019 14:30, amylumey2005
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2


Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
Do you know the correct answer?
Dihydrogen sulfide reacts with oxygen gas to form sulfur dioxide and water. How many moles of oxygen...
Questions in other subjects:


Mathematics, 04.04.2020 18:23

Physics, 04.04.2020 18:23

Mathematics, 04.04.2020 18:23

Mathematics, 04.04.2020 18:23

Geography, 04.04.2020 18:23


Social Studies, 04.04.2020 18:23


Mathematics, 04.04.2020 18:23


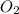 will react when 0.6469 moles of water are formed.
will react when 0.6469 moles of water are formed.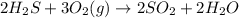
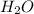 are formed from = 3 moles of
are formed from = 3 moles of 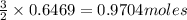 of
of 



