
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxide gas, NO(g). This step is represented by the following balanced equation: 4 NH3(g) + 5 O2(g) --> 4 NO(g) + 6 H2O(l) Determine the amount of oxygen needed to produce 1.2 x 104 mol of nitrogen monoxide gas.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 19:10, PineaPPle663
Nuclear fusion is the source of energy for stars. besides hydrogen, which other element is most likely also common in stars?
Answers: 1

Chemistry, 22.06.2019 08:30, ebigham5117
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2
Do you know the correct answer?
One essential step in the manufacture of many plant fertilizers is the production of nitrogen monoxi...
Questions in other subjects:

Mathematics, 21.08.2019 04:30



History, 21.08.2019 04:30

Mathematics, 21.08.2019 04:30






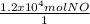 ×
× 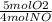 = 1.5 moles of O2 needed
= 1.5 moles of O2 needed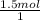 ×
× 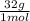 = 48 grams needed
= 48 grams needed



