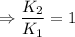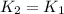Chemistry, 09.03.2021 04:40, blancademarco1994
2) A common "rule of thumb" -- for many reactions around room temperature is that the
rate will double for each ten degree increase in temperature. Does the reaction you hav
studied seem to obey this rule.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2


Chemistry, 22.06.2019 11:50, tajanaewilliams77
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3
Do you know the correct answer?
2) A common "rule of thumb" -- for many reactions around room temperature is that the
rate will d...
Questions in other subjects:

Mathematics, 07.01.2020 04:31


History, 07.01.2020 04:31






Social Studies, 07.01.2020 04:31

Mathematics, 07.01.2020 04:31


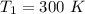
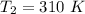
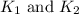 respectively.
respectively.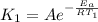 and
and 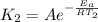
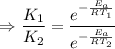
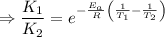
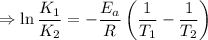
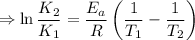
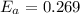 J/mol
J/mol