
Chemistry, 09.03.2021 02:50, quincyjosiah07
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs out 32.mg of oxalic acid H2C2O4, a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in 250.mL of distilled water. The student then titrates the oxalic acid solution with his sodium hydroxide solution. When the titration reaches the equivalence point, the student finds he has used 60.9mL of sodium hydroxide solution. Calculate the molarity of the student's sodium hydroxide solution. Round your answer to 2 significant digits. M

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, gatorr2010
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 10:30, perezanthony2403
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 13:00, nadikadiaz1
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1
Do you know the correct answer?
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs o...
Questions in other subjects:





Computers and Technology, 15.12.2020 21:50

Mathematics, 15.12.2020 21:50

History, 15.12.2020 21:50




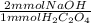 = 0.72 mmol NaOH
= 0.72 mmol NaOH



