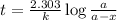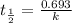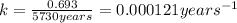Chemistry, 08.03.2021 23:10, robert7248
The previous part could be done without using the decay equation, because the ratio of original 14C14C to present 14C14C was an integer power of 1/2. Most problems are not so simple. To solve more general carbon-dating problems, you must first find the value of the decay constant for 14C14C, so that you can easily use the decay equation. Using the given half-life, 5730 yearsyears, find the value of the decay constant for 14C14C. Express your answer in inverse years to three significant figures. View Available Hint(s)

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:30, drivinghydra
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1


Chemistry, 22.06.2019 12:40, carebear60
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1
Do you know the correct answer?
The previous part could be done without using the decay equation, because the ratio of original 14C1...
Questions in other subjects:


Mathematics, 30.01.2021 21:40

Advanced Placement (AP), 30.01.2021 21:40

Biology, 30.01.2021 21:40

Mathematics, 30.01.2021 21:40



Mathematics, 30.01.2021 21:40

Mathematics, 30.01.2021 21:40

Mathematics, 30.01.2021 21:40