
Please help , don’t answer if u don’t know please
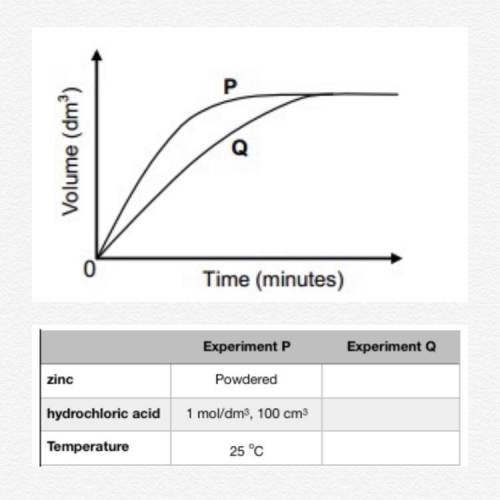
An experiment is carried out to investigate the rate at which hydrogen gas is produced when a given amount of zinc reacts with an excess of a dilute hydrochloric acid solution. Curve P on the graph below shows the volume of hydrogen gas formed when 100 cm3 of a 1 mol/dm3 solution is used. (Reaction rate is indicated by the slope of the graphs.)
Zn + 2 HCl = ZnCl2 + H2
a. Design an experiment that would produce curve Q on the graph below. (Hint: change the experimental parameters so that the rate of gas evolution changes according to the curves). You can also use the table to record your data.
b. What is the implication of the fact that the two curves touch? Explain.
c. Give a reason why the rate of hydrogen production slows down towards the end in both experiments.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, kevinh2683
Apump contains 0.5 l of air at 203 kpa. you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 17:30, rollercoasterbuddies
Why is the melting of ice a physical change ?
Answers: 1
Do you know the correct answer?
Please help , don’t answer if u don’t know please
An experiment is carried out to investigate the r...
Questions in other subjects:

Social Studies, 23.07.2019 12:00


Mathematics, 23.07.2019 12:00


Mathematics, 23.07.2019 12:00



English, 23.07.2019 12:00


Biology, 23.07.2019 12:00






