
Chemistry, 05.03.2021 19:00, sydneepollitt32
Carbon dioxide and water react to form methanol and oxygen, like this:
2CO2(g) + 4H2O(g) → 2CH3OH(l) + 3O2 (g)
At a certain temperature, a chemist finds that a 7.5L reaction vessel containing a mixture of carbon dioxide, water, methanol, and oxygen at equilibrium has the following composition:
Compound Amount
CO2 3.28g
H2O 3.86g
CH3OH 1.51g
O2 2.80g
Required:
Calculate the value of the equilibrium constant Kc for this reaction. Round your answer to 2 significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:10, mistiehaas
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2



Chemistry, 22.06.2019 14:20, greenbyron88
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
Do you know the correct answer?
Carbon dioxide and water react to form methanol and oxygen, like this:
2CO2(g) + 4H2O(g) → 2CH3OH(l...
Questions in other subjects:






Mathematics, 11.12.2020 01:00

Computers and Technology, 11.12.2020 01:00

English, 11.12.2020 01:00

History, 11.12.2020 01:00


![\frac{[O_2]^3}{[CO_2]^2[H_2O]^4}](/tpl/images/1172/1518/03065.png)
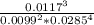 = 24x10³
= 24x10³



