
Chemistry, 20.09.2019 19:00, arthurdolz
For the reaction shown, calculate how many grams of oxygen form when each quantity of reactant completely reacts.
2 kclo3(s) → 2 kcl(s) + 3 o2(g)
a. 2.72 g kclo3
b. 0.361 g kclo3
c. 83.6 kg kclo3
d. 22.5 mg kclo3

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, terrancebest
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2

Chemistry, 22.06.2019 04:30, anthony4034
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 13:00, rome58
Lab reagent, hypothesis test. a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl. these six measurements are assumed to be an srs of all possible measurements from solution. they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution. carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
Do you know the correct answer?
For the reaction shown, calculate how many grams of oxygen form when each quantity of reactant compl...
Questions in other subjects:




English, 10.12.2020 01:10





Arts, 10.12.2020 01:10


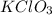 = 39.1 + 35.5 + 3(16.0) = 122.6 g
= 39.1 + 35.5 + 3(16.0) = 122.6 g = 32.0 g
= 32.0 g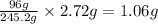 of
of 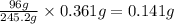 of
of 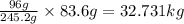 of
of 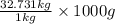 = 32731 g of
= 32731 g of 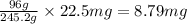
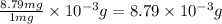 of
of 



