
Chemistry, 02.03.2021 21:20, theblackdot16
an unknown sample with a mass of 50.0 grams changes from an initial temperature of 22.5°C to a final temperature of 32.4°C. It is known that the sample absorbed 444 J and f heat l. Bases on this information, what substance in the table is most likely the identity of this sample?
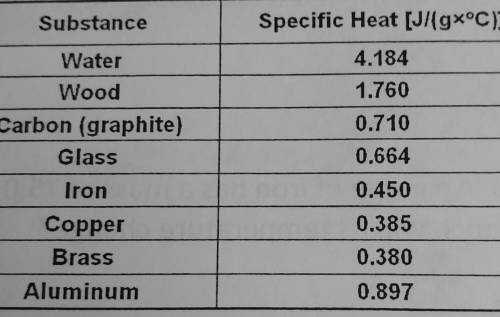

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 03:00, duplessistoccara
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 2
Do you know the correct answer?
an unknown sample with a mass of 50.0 grams changes from an initial temperature of 22.5°C to a final...
Questions in other subjects:

Mathematics, 03.12.2020 07:10


Chemistry, 03.12.2020 07:10

Biology, 03.12.2020 07:10



Mathematics, 03.12.2020 07:10

Physics, 03.12.2020 07:10








