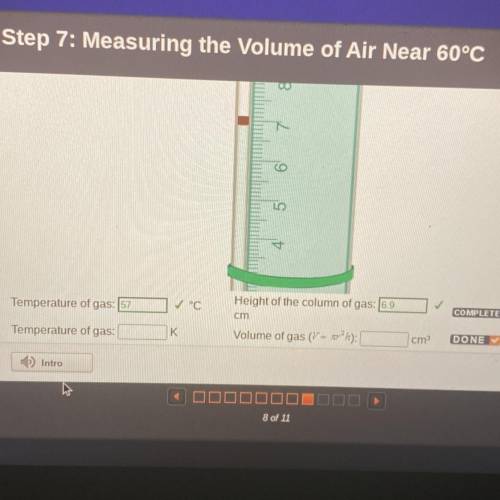
Temperature of gas: 57
✓ °C
Height of the column of gas: 6.9
COMPLETE
cm
Te...

Chemistry, 02.03.2021 06:00, hannahsambrano12
Temperature of gas: 57
✓ °C
Height of the column of gas: 6.9
COMPLETE
cm
Temperature of gas:
K
Volume of gas (V = xr2 ha):
cm
DONE

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 17:30, tiffanyhmptn
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 21:30, djdjdjdbdbjx
What is another way to determine mass times acceleration?
Answers: 1
Do you know the correct answer?
Questions in other subjects:

English, 19.01.2020 22:31







Mathematics, 19.01.2020 22:31

Mathematics, 19.01.2020 22:31

Mathematics, 19.01.2020 22:31






