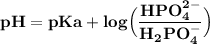Chemistry, 02.03.2021 04:50, germainenez3288
On the basis of the information above, a buffer with a pH = 9 can best be made by using
A pure NaH2PO4
B H3PO4 + H2PO4–
C H2PO4– + PO42–
D H2PO4– + HPO42–
E HPO42– + PO43

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 03:10, mani1682
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2


Chemistry, 23.06.2019 15:00, ericperkins10ox0b27
In two or more complete sentences describe all of the van der waals forces that exist between molecules of sulfur dioxide, so2.
Answers: 1
Do you know the correct answer?
On the basis of the information above, a buffer with a pH = 9 can best be made by using
A pure NaH2...
Questions in other subjects:




Biology, 10.03.2021 03:50


Physics, 10.03.2021 03:50

Mathematics, 10.03.2021 03:50

Mathematics, 10.03.2021 03:50



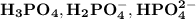 are
are 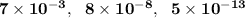 respectively.
respectively.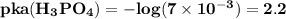
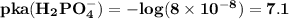
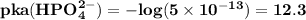
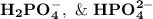 lies on either side of the desired pH of the buffer. This implies that one is slightly over and the other is slightly under.
lies on either side of the desired pH of the buffer. This implies that one is slightly over and the other is slightly under.