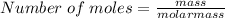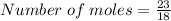Chemistry, 02.03.2021 03:00, emilyplays474
I have calculated 23 grams of water in a laboratory. What is the amount of molecules
present in this amount?
a. None of the above
b. 8.3x10^24 molecules
c. 6.9x10^23 molecules
d. 7.7x10^23 molecules
e. 5.6x10^25 molecules

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, mbrisen7420
Compare and contrast physical changes with chemical changes.
Answers: 3


Chemistry, 22.06.2019 10:00, paynedeforest2596
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 22:30, angelagonzalesownus1
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1
Do you know the correct answer?
I have calculated 23 grams of water in a laboratory. What is the amount of molecules
present in thi...
Questions in other subjects:

Mathematics, 04.08.2019 10:00

History, 04.08.2019 10:00


Mathematics, 04.08.2019 10:00

Biology, 04.08.2019 10:00



History, 04.08.2019 10:00

Chemistry, 04.08.2019 10:00

Mathematics, 04.08.2019 10:00