
Chemistry, 02.03.2021 01:00, smartcookie8251
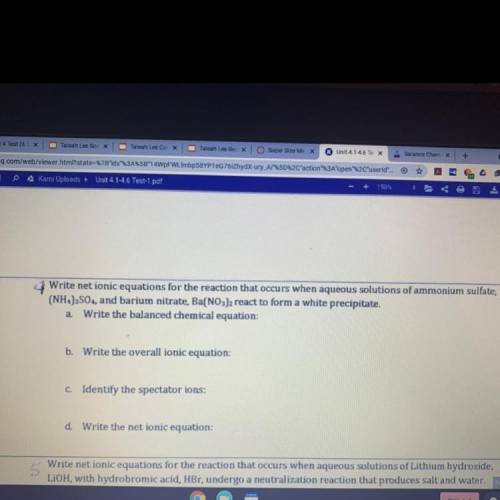
Write net ionic equations for the reaction that occurs when aqueous solutions of ammonium sulfate
(NH4)2SO,, and barium nitrate, Ba(NO3)2 react to form a white precipitate.
a Write the balanced chemical equation:
b. Write the overall ionic equation:
C. Identify the spectator ions:
d Write the net ionic equation:

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 18:00, kamjay2006
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1

Chemistry, 23.06.2019 04:31, woodfordmaliky
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1
Do you know the correct answer?
Write net ionic equations for the reaction that occurs when aqueous solutions of ammonium sulfate
(...
Questions in other subjects:

Chemistry, 04.06.2021 05:20


Mathematics, 04.06.2021 05:20

Mathematics, 04.06.2021 05:20

Mathematics, 04.06.2021 05:20

Mathematics, 04.06.2021 05:20


Chemistry, 04.06.2021 05:20








