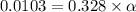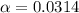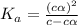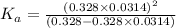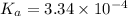Chemistry, 01.03.2021 22:20, william1690
In the laboratory, a general chemistry student measured the pH of a 0.328 M aqueous solution of acetylsalicylic acid (aspirin), HC9H7O4 to be 1.987. Use the information she obtained to determine the Ka for this acid.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, huangjianhe135
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 04:00, clairebear66
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1
Do you know the correct answer?
In the laboratory, a general chemistry student measured the pH of a 0.328 M aqueous solution of acet...
Questions in other subjects:




Physics, 21.04.2020 17:46

Mathematics, 21.04.2020 17:46




Mathematics, 21.04.2020 17:46


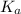 for the acid is
for the acid is 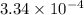
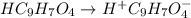
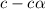
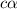
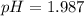
![1.987=-log[H^+]](/tpl/images/1158/1923/8f2fb.png)
![[H^+]=0.0103](/tpl/images/1158/1923/4a7f1.png)
![[H^+]=c\times \alpha](/tpl/images/1158/1923/4fc41.png)