Chemistry, 01.03.2021 21:50, nocomprendoplshelp
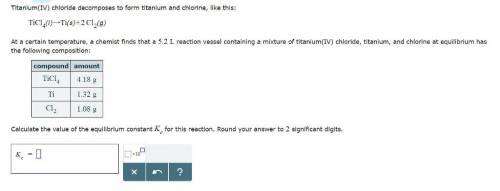
Titanium(IV) chloride decomposes to form titanium and chlorine, like this:
TiCl4(l) → Ti(s)+ 2Cl2(g)
At a certain temperature, a chemist finds that a reaction vessel containing a mixture of titanium(IV) chloride, titanium, and chlorine at equilibrium has the following composition:
Compound Amount
TiCl4 4.18g
Ti 1.32g
Cl2 1.08g
Required:
Calculate the value of the equilibrium constant for this reaction.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:30, vhife4901
Which of these statements best explains why space exploration should be encouraged? it prepares humans to live without oxygen. it dispel myths about objects in space. it prevents comets and asteroids from striking earth. it creates technology to absorb harmful radiations in space.
Answers: 1

Chemistry, 23.06.2019 03:00, makayyafreeman
Select the correct answer. wax is a nonpolar substance. in which type of substance is it the most soluble?
Answers: 1
Do you know the correct answer?
Titanium(IV) chloride decomposes to form titanium and chlorine, like this:
TiCl4(l) → Ti(s)+ 2Cl2(g...
Questions in other subjects:








Mathematics, 19.03.2021 20:00


Mathematics, 19.03.2021 20:00