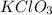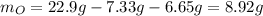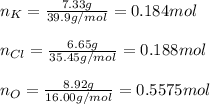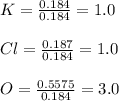Chemistry, 01.03.2021 08:50, noreenhussain
Help?
A 22.98 g sample of a compound contains 7.33 g of potassium, K, 6.65 g of chlorine, Cl, and oxygen, O. Calculate the empirical formula.
Insert subscripts as needed.
Empirical formula: KClO

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, isabelvaldez123
The boiling point of liquids is very high what does it indicate
Answers: 1


Chemistry, 22.06.2019 15:00, hockeykid7583
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 22:30, xlebrny7831
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
Do you know the correct answer?
Help?
A 22.98 g sample of a compound contains 7.33 g of potassium, K, 6.65 g of chlorine, Cl, and o...
Questions in other subjects:



Mathematics, 30.04.2021 20:50






Mathematics, 30.04.2021 20:50

English, 30.04.2021 20:50