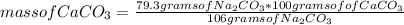Chemistry, 28.02.2021 23:40, haileyhale5
For the reaction Na2CO3+Ca(NO3)2⟶CaCO3+2NaNO3 how many grams of calcium carbonate, CaCO3, are produced from 79.3 g of sodium carbonate?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 01:00, stefaniethibodeaux
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1

Chemistry, 23.06.2019 11:00, artiomtyler007
Find the enthalpy of neutralization of hcl and naoh. 87 cm3 of 1.6 mol dm-3 hydrochloric acid was neutralized by 87 cm3 of 1.6 mol dm-3 naoh. the temperature rose from 298 k to 317.4 k. the specific heat capacity is the same as water, 4.18 j/k g. a. -101.37 kj b. 7055 kj c. 10,1365 kj
Answers: 1
Do you know the correct answer?
For the reaction Na2CO3+Ca(NO3)2⟶CaCO3+2NaNO3 how many grams of calcium carbonate, CaCO3, are produc...
Questions in other subjects:

Mathematics, 18.05.2021 21:50

Mathematics, 18.05.2021 21:50

History, 18.05.2021 21:50



History, 18.05.2021 21:50



Mathematics, 18.05.2021 21:50