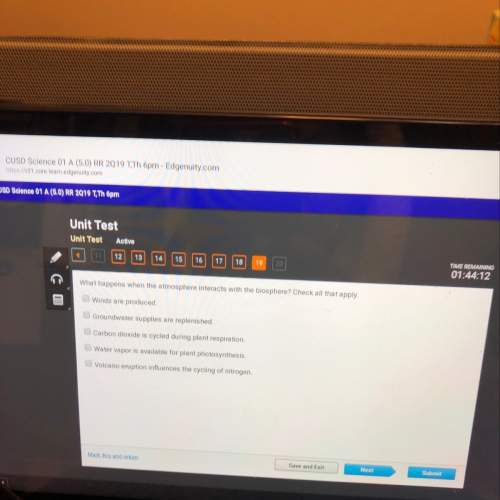
Chemistry, 27.02.2021 01:00, ezellcarly
When ethane (C2H6) burns, it produces carbon dioxide and water:
2C2H6(g)+7O2(g)→4CO2(g)+6H2O(l)
How many moles of water will be produced when 3 moles of ethane are burned?
3 moles
6 moles
9 moles
12 moles

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2

Chemistry, 22.06.2019 14:20, kekecantonxox121
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 20:30, demarcuswiseman
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
Do you know the correct answer?
When ethane (C2H6) burns, it produces carbon dioxide and water:
2C2H6(g)+7O2(g)→4CO2(g)+6H2O(l)
Questions in other subjects:

Biology, 05.01.2020 23:31









Mathematics, 05.01.2020 23:31