
Chemistry, 26.02.2021 20:40, Brangolflro
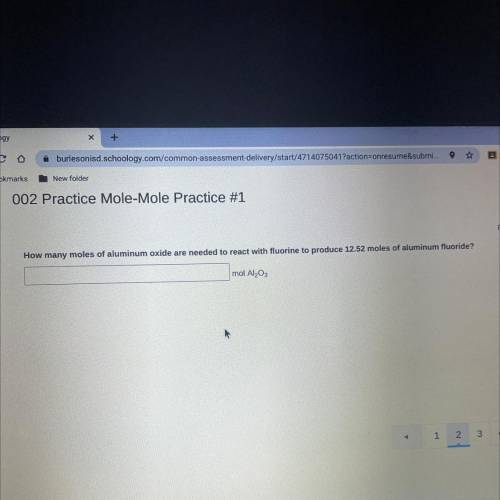
How many moles of aluminum oxide are needed to react fluorine to produce 12.52 moles of aluminum fluoride

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, allyyzz
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3


Chemistry, 23.06.2019 02:00, hermesrobles
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2

Do you know the correct answer?
How many moles of aluminum oxide are needed to react fluorine to produce 12.52 moles of aluminum flu...
Questions in other subjects:



Mathematics, 21.09.2021 01:40

Mathematics, 21.09.2021 01:40


Mathematics, 21.09.2021 01:40

Mathematics, 21.09.2021 01:40


Biology, 21.09.2021 01:40

Mathematics, 21.09.2021 01:40






