

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, uniqueray33
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 10:00, sdlesley66
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1


Chemistry, 23.06.2019 13:20, dani9427
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. this reaction is now the first step taken to make most of the world's fertilizer. suppose a chemical engineer studying a new catalyst for the haber reaction finds that 671 liters per second of dinitrogen are consumed when the reaction is run at 271c and 0.99atm. calculate the rate at which ammonia is being produced. give your answer in kilograms per second. round your answer to significant digits.
Answers: 3
Do you know the correct answer?
The compound [Fe(SCN)(OH2 )5 ] 2 can be detected in the reaction of [Co(NCS)(NH3 )5 ]2 with Fe2 (aq)...
Questions in other subjects:


English, 31.01.2021 03:30

Mathematics, 31.01.2021 03:30



Mathematics, 31.01.2021 03:30





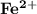 since it is an oxidizing agent that produces
since it is an oxidizing agent that produces  complex in solution.
complex in solution.![\mathbf{[Co(NH_3)_5(NCS)]^{+2}+[Fe(H_2O)_6]^{+2} \to [Fe(SCN) (H_2O)_5]^{+2}+ [Co(H_2O)_6]^{+2}}](/tpl/images/1147/8875/ef449.png)




