

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:00, teacherpreacher
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2



Chemistry, 23.06.2019 03:00, amberskids2
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
Do you know the correct answer?
In a COMPLETE PARAGRAPH, describe the procedure to calculating the mass of a produce produced in a c...
Questions in other subjects:

English, 20.08.2019 00:30

Mathematics, 20.08.2019 00:30


Mathematics, 20.08.2019 00:30

History, 20.08.2019 00:30




Mathematics, 20.08.2019 00:30




 :
: 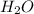 = 1: 2
= 1: 2 of
of 



