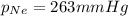A 2.00L mixture of helium, nitrogen, and neon has a total pressure of 815 mmHg at a
temperature of 255K. If the partial pressure of helium is 201 mmHg and the partial
pressure of nitrogen is 351 mmHg, what is the partial pressure of neon in the mixture?
O 709 mmHg
O 512 mmHg
O 667 mmHg
O 263 mmHg

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, jasmineharris121
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1



Chemistry, 23.06.2019 00:30, kylee65
What are the advantages of using the metric system? designed as a decimal system making conversions simpler more accurate system of measurement has prefixes that correspond to an amount to use with all base units used by the entire scientific community
Answers: 2
Do you know the correct answer?
A 2.00L mixture of helium, nitrogen, and neon has a total pressure of 815 mmHg at a
temperature of...
Questions in other subjects:


History, 25.12.2021 18:00


Mathematics, 25.12.2021 18:00

English, 25.12.2021 18:00

Biology, 25.12.2021 18:00


Mathematics, 25.12.2021 18:00



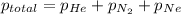
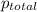 =total pressure of gases = 815 mm Hg
=total pressure of gases = 815 mm Hg
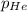 = partial pressure of helium = 201 mm Hg
= partial pressure of helium = 201 mm Hg
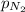 = partial pressure of nitrogen = 351 mm Hg
= partial pressure of nitrogen = 351 mm Hg
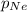 = partial pressure of Neon = ?
= partial pressure of Neon = ?