
Chemistry, 23.02.2021 21:10, deanlmartin
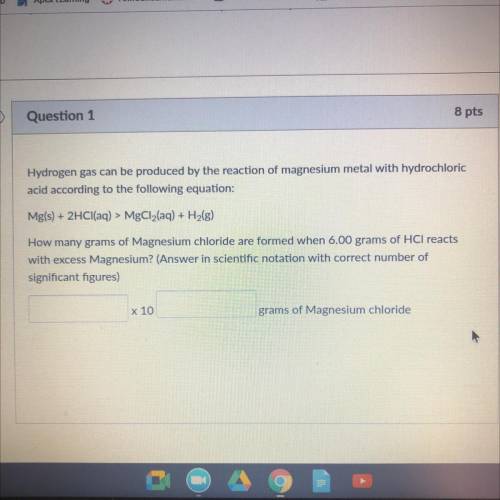
Hydrogen gas can be produced by the reaction of magnesium metal WICH
acid according to the following equation:
Mg(s) + 2HCl(aq) > MgCl2(aq) + H2(g)
How many grams of Magnesium chloride are Irmed when 6.00 grams of HCl reacts
with excess Magnesium? (Answer in scientific notation with correct number of
significant figures)
x 10
grams of Magnesium chloride

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, brookemcelhaney
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1


Chemistry, 22.06.2019 12:30, robert7248
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2
Do you know the correct answer?
Hydrogen gas can be produced by the reaction of magnesium metal WICH
acid according to the followin...
Questions in other subjects:

History, 06.10.2019 10:20


History, 06.10.2019 10:20

Physics, 06.10.2019 10:20


Physics, 06.10.2019 10:20

Mathematics, 06.10.2019 10:20








