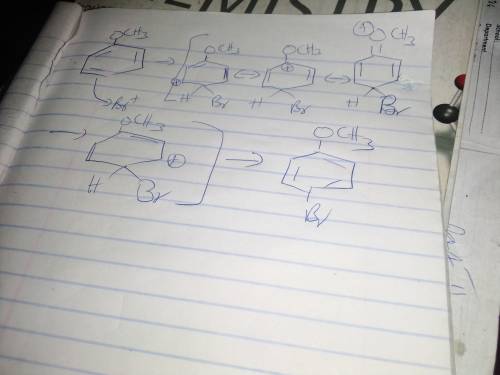
Chemistry, 22.02.2021 19:50, villarrealc1987
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Groups, already present on the benzene ring, that direct ortho/para further stabilize this intermediate by participating in the resonance delocalization of the positive charge. Assume that the following group is present on a benzene ring at position 1 and that you are brominating the ring at positon 4. In the box below draw the structure of the resonance contributor that shows this group actively participating in the charge delocalization. -OCH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:20, rscvsdfsrysas1857
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 21:30, rileydavidharless
Which substance can be broken down by chemical means
Answers: 1
Do you know the correct answer?
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Gr...
Questions in other subjects:

Biology, 17.12.2019 03:31

Mathematics, 17.12.2019 03:31


Mathematics, 17.12.2019 03:31

Mathematics, 17.12.2019 03:31

Biology, 17.12.2019 03:31


Mathematics, 17.12.2019 03:31

Social Studies, 17.12.2019 03:31