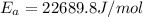Chemistry, 22.02.2021 18:40, danbat3023
The rate constant for a certain reaction is measured at two different temperatures:
temperature k
376.0 °C 4.8 x 10^8
280.0 °C 2.3 x 10^8
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy Ea for this reaction.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, lasagnafoe
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 12:30, masteroftheuniverse3
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2

Do you know the correct answer?
The rate constant for a certain reaction is measured at two different temperatures:
temperature k
Questions in other subjects:



Chemistry, 04.04.2020 07:31





Mathematics, 04.04.2020 07:31



![ln \frac{k_{2}}{k_{1}} = \frac{-E_{a}}{R}[\frac{1}{T_{2}} - \frac{1}{T_{1}}]](/tpl/images/1136/3217/c502b.png)
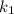 = rate constant at temperature
= rate constant at temperature 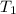 =
= 
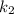 = rate constant at temperature
= rate constant at temperature 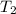 =
= 
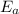 = activation energy = ?
= activation energy = ?
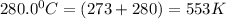
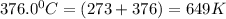
![ln \frac{4.8\times 10^8}{2.3\times 10^8} = \frac{-E_{a}}{8.314}[\frac{1}{649} - \frac{1}{553}]](/tpl/images/1136/3217/fcab8.png)