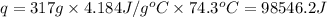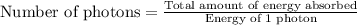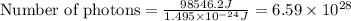Chemistry, 28.12.2019 18:31, stophendless9780
Acontainer with 0.317 l of water is placed into microwave and is then radiated with electromagnetic energy that has a wavelength of 13.3 cm. the temperature of the water then rose by 74.3 °c. calculate the number of photons that were absorbed by the water. assume water has a density of 1.00 g·ml–1 and its specific heat is 4.184 j·g–1·°c–1

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, bernicewhite156
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 02:00, hemolelekeakua
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Do you know the correct answer?
Acontainer with 0.317 l of water is placed into microwave and is then radiated with electromagnetic...
Questions in other subjects:


SAT, 30.01.2020 20:51


Mathematics, 30.01.2020 20:51

Mathematics, 30.01.2020 20:51

Mathematics, 30.01.2020 20:51

English, 30.01.2020 20:51

Computers and Technology, 30.01.2020 20:51



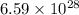
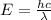
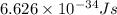
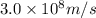
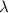 = wavelength of photon = 13.3 cm = 0.133 m (Conversion factor: 1 m = 100 cm )
= wavelength of photon = 13.3 cm = 0.133 m (Conversion factor: 1 m = 100 cm )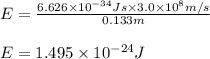
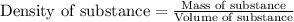
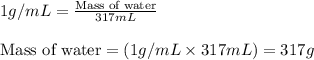
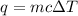
 = change in temperature = 74.3°C
= change in temperature = 74.3°C