
Chemistry, 19.02.2021 23:40, destineepreuss4472
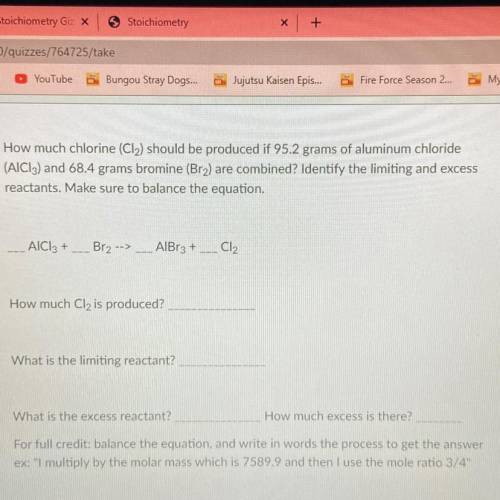
⚠️PLZ HELP TIMED TEST⚠️How much chlorine (Cl2) should be produced if 95.2 grams of aluminum chloride
(AIC13) and 68.4 grams bromine (Bra) are combined? Identify the limiting and excess
reactants. Make sure to balance the equation.
AlCl3 +_ Br2 -->
AlBr3 +
Cl2
How much Cl2 is produced?
What is the limiting reactant?
What is the excess reactant?
How much excess is there?
For full credit: balance the equation, and write in words the process to get the answer
ex: "I multiply by the molar mass which is 7589.9 and then I use the mole ratio 3/4"

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:00, ghernadez
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1


Do you know the correct answer?
⚠️PLZ HELP TIMED TEST⚠️How much chlorine (Cl2) should be produced if 95.2 grams of aluminum chloride...
Questions in other subjects:


Business, 19.01.2022 02:30

Geography, 19.01.2022 02:30



Mathematics, 19.01.2022 02:30









