
Chemistry, 19.02.2021 06:10, yasameenakbari
What is the molecular formula for a compound that is 44.87% potassium, 36.7% oxygen, 18.0% sulfur and a molecular mass of 696g.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:40, kellymcdow5135
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Do you know the correct answer?
What is the molecular formula for a compound that is 44.87% potassium, 36.7%
oxygen, 18.0% sulfur a...
Questions in other subjects:



Biology, 27.01.2021 20:00

Mathematics, 27.01.2021 20:00


Mathematics, 27.01.2021 20:00

English, 27.01.2021 20:00

Physics, 27.01.2021 20:00


Business, 27.01.2021 20:00


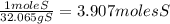
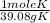 = 7.99117 mole K
= 7.99117 mole K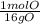 = 15.9654 mole O
= 15.9654 mole O



