
Chemistry, 17.02.2021 01:00, maritzahernandez32
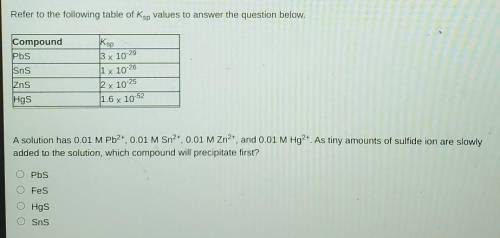
Refer to the following table of Ksp values to answer the question below. Compound
A solution has 0.01 M Pb2+, 0.01 M Sn2+, 0.01 M Zn2+, and 0.01 M Hg2+. As tiny amounts of sulfide ion are slowly added to the solution, which compound will precipitate first?
A. Pbs
B. Fes
C. Hgs
D. Sns

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:10, akatsionis25
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2


Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
Do you know the correct answer?
Refer to the following table of Ksp values to answer the question below. Compound
A solution has 0...
Questions in other subjects:

Mathematics, 14.01.2020 05:31

Chemistry, 14.01.2020 05:31




Mathematics, 14.01.2020 05:31

English, 14.01.2020 05:31

Mathematics, 14.01.2020 05:31


Biology, 14.01.2020 05:31






