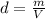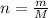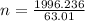Chemistry, 14.02.2021 14:00, patrick171888
A nitric acid solution containing 71.0% HNO3 (by mass) is available in a laboratory. How many moles of HNO3 are present in 1.98 L of this solution?
a.
62.8 mol
b.
31.7 mol
c.
22.3 mol
d.
0.0316 mol
e.
none of these

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:40, alexisbcatlett14
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2


Chemistry, 23.06.2019 10:30, fatheadd2007
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
Do you know the correct answer?
A nitric acid solution containing 71.0% HNO3 (by mass) is available in a laboratory. How many moles...
Questions in other subjects:



Mathematics, 03.11.2020 20:30

History, 03.11.2020 20:30

Social Studies, 03.11.2020 20:30

Computers and Technology, 03.11.2020 20:30

English, 03.11.2020 20:30


Health, 03.11.2020 20:30

Mathematics, 03.11.2020 20:30