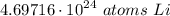Chemistry, 14.02.2021 01:00, SavannahP04
Calculate the number of Li atoms in 7.8 mol of Li. Express your answer using two significant figures.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, ggdvj9gggsc
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1

Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 22.06.2019 23:30, treylartigue
The appropriate concentration for an iodine sanitizer is
Answers: 1

Chemistry, 23.06.2019 03:10, 3jazybraxy
Which is true according to the law of conservation of energy
Answers: 1
Do you know the correct answer?
Calculate the number of Li atoms in 7.8 mol of Li.
Express your answer using two significant figure...
Questions in other subjects:



Mathematics, 18.11.2020 23:40

Mathematics, 18.11.2020 23:40


English, 18.11.2020 23:40

Geography, 18.11.2020 23:40

Mathematics, 18.11.2020 23:40


Advanced Placement (AP), 18.11.2020 23:40


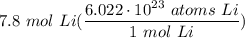 Multiply/Divide:
Multiply/Divide: