
Chemistry, 12.02.2021 22:20, brookeleneewick
Three Anacharis plants were placed inside test tubes filled with water. once test was exposed to low light, one to medium light, and another was exposed to high light. the table shows the results of experiment. based on the information shown, what was the difference in volume (in ml) of oxygen produced between the low light plants and high light plants after 45 minutes?
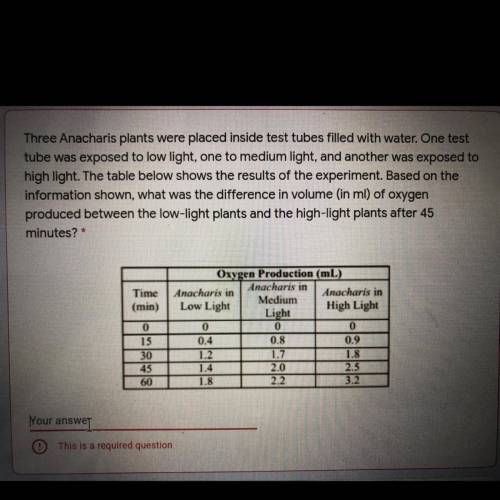

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 23:40, sydneykated
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
Do you know the correct answer?
Three Anacharis plants were placed inside test tubes filled with water. once test was exposed to low...
Questions in other subjects:



Biology, 31.01.2020 04:49


Health, 31.01.2020 04:49




Mathematics, 31.01.2020 04:50

Mathematics, 31.01.2020 04:50






