
Chemistry, 12.02.2021 08:40, asjessehills2000
The monovalent salt concentration (the predominant solute in the blood cell) for a sample of red blood cells is 0.13 moles/liter. If one of these red blood cells were placed in pure water (at around room temperature, 300 K), and the cell comes to hydrostatic equilibrium with the water, what is the osmotic pressure of the cell (assuming it doesn't burst)

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:30, tateandvioletAHS14AY
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
Do you know the correct answer?
The monovalent salt concentration (the predominant solute in the blood cell) for a sample of red blo...
Questions in other subjects:

English, 29.09.2020 21:01




Mathematics, 29.09.2020 21:01


Mathematics, 29.09.2020 21:01




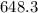 KPa
KPa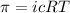
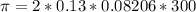
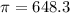 KPa
KPa



