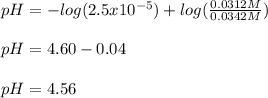Use the Henderson-Hasselbalch equation, eq. (3), to calculate the pH expected for a buffer solution prepared from this acid and its conjugate base, assuming: the mass of the weak acid is 0.2 g and the mass of the conjugate base is 0.2 g will be added to 25 mL of water. Also assume the molar mass of the weak acid is 234 and its conjugate base is the sodium salt.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
Do you know the correct answer?
Use the Henderson-Hasselbalch equation, eq. (3), to calculate the pH expected for a buffer solution...
Questions in other subjects:

Chemistry, 21.07.2019 16:30



Physics, 21.07.2019 16:30







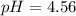
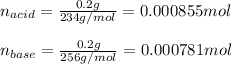
![[acid]=0.000855mol/0.025L=0.0342M](/tpl/images/1113/0020/c5975.png)
![[base]=0.000781mol/0.025L=0.0312M](/tpl/images/1113/0020/790f7.png)