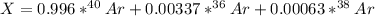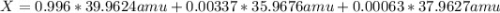Argon has three naturally-occurring isotopes: 99.6% of 40Ar, with an atomic weight of 39.9624 amu, 0.337% of 36Ar, with an atomic weight of 35.9676 amu, and 0.063% of 38Ar, with an atomic weight of 37.9627 amu. Calculate the average atomic weight of Ar. Round off the answer to six significant figures. Do not include the units.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 14:30, Dreynolds1667
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1
Do you know the correct answer?
Argon has three naturally-occurring isotopes: 99.6% of 40Ar, with an atomic weight of 39.9624 amu, 0...
Questions in other subjects:


Mathematics, 05.11.2020 19:50

Mathematics, 05.11.2020 19:50

Mathematics, 05.11.2020 19:50


English, 05.11.2020 19:50

Biology, 05.11.2020 19:50

Chemistry, 05.11.2020 19:50


Biology, 05.11.2020 19:50