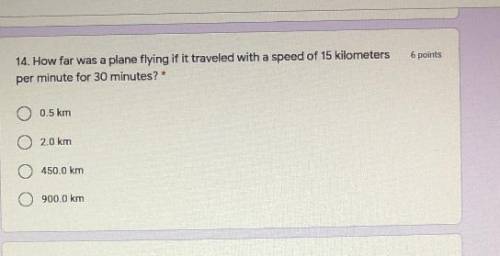
Help ASAP I will mark brainliest for the correct answer !!!⚡️⚡️⚡️
...

Chemistry, 11.02.2021 16:40, danielobanoyen
Help ASAP I will mark brainliest for the correct answer !!!⚡️⚡️⚡️

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:10, roserose3098
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
Do you know the correct answer?
Questions in other subjects:




English, 12.08.2020 04:01


Mathematics, 12.08.2020 04:01


Mathematics, 12.08.2020 04:01


Physics, 12.08.2020 04:01







