
Chemistry, 10.02.2021 23:50, reaperqueen21
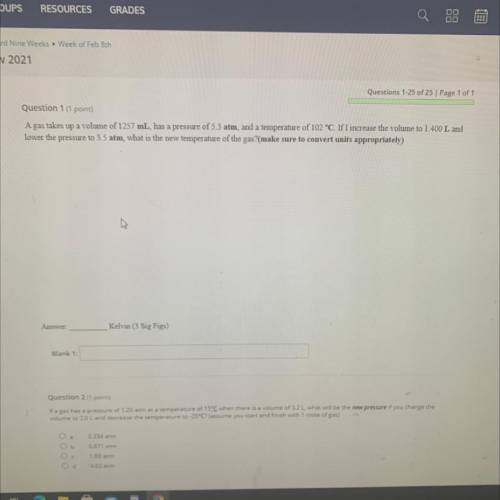
A gas takes up a volume of 1257 mL, has a pressure of 5.3 atm, and a temperature of 102 °C. If I increase the volume to 1.400 L and
lower the pressure to 3.5 atm, what is the new temperature of the gas?(make sure to convert units appropriately)
Kelvin (3 Sig Figs)
Blank 1:

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, genyjoannerubiera
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 21:00, ciel8809
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
Do you know the correct answer?
A gas takes up a volume of 1257 mL, has a pressure of 5.3 atm, and a temperature of 102 °C. If I inc...
Questions in other subjects:






Mathematics, 27.08.2020 23:01




Mathematics, 27.08.2020 23:01







