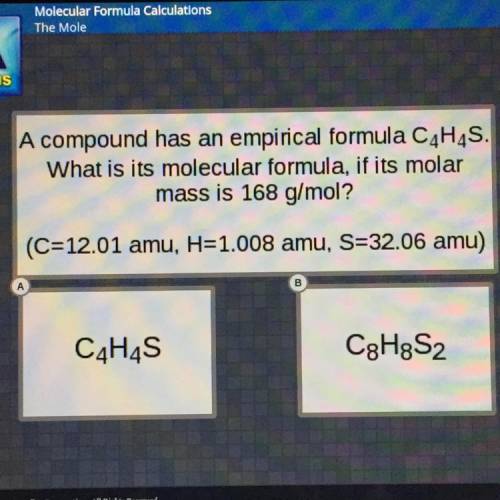
A compound has an empirical formula C4H4S.
What is its molecular formula, if its molar
mass i...

Chemistry, 09.02.2021 01:00, natalyarenassalgado
A compound has an empirical formula C4H4S.
What is its molecular formula, if its molar
mass is 168 g/mol?
(C=12.01 amu, H=1.008 amu, S=32.06 amu)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, codeyhatch142
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 00:00, nyasiasaunders1234
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 03:00, Dkhaurithompson
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1
Do you know the correct answer?
Questions in other subjects:





Chemistry, 04.08.2019 04:30


Social Studies, 04.08.2019 04:30

Biology, 04.08.2019 04:30


Physics, 04.08.2019 04:30






