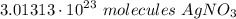Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, DarcieMATHlin2589
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1


Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 06:00, citlalli30
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2
Do you know the correct answer?
How many molecules are in 85g of silver nitrate?...
Questions in other subjects:

Mathematics, 25.09.2019 20:00

Biology, 25.09.2019 20:00

Mathematics, 25.09.2019 20:00

Social Studies, 25.09.2019 20:00

Mathematics, 25.09.2019 20:00


Social Studies, 25.09.2019 20:00

Mathematics, 25.09.2019 20:00

Social Studies, 25.09.2019 20:00


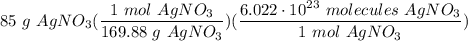 Multiply/Divide:
Multiply/Divide: