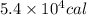Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1


Chemistry, 22.06.2019 21:50, namoralessimon03
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
Do you know the correct answer?
How much heat (in calories) must be applied to a 100 ml beaker filled with boiling water in order to...
Questions in other subjects:

Mathematics, 25.10.2019 20:43

History, 25.10.2019 20:43

Mathematics, 25.10.2019 20:43

Physics, 25.10.2019 20:43

Biology, 25.10.2019 20:43

Mathematics, 25.10.2019 20:43


Mathematics, 25.10.2019 20:43

Mathematics, 25.10.2019 20:43

Physics, 25.10.2019 20:43