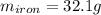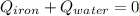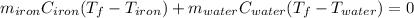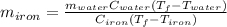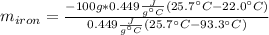Chemistry, 05.02.2021 21:20, eeeeee7891
A sample of iron, which has a specific heat capacity of 0.449 Jg^-1℃^-1, is put into a calorimeter (see sketch at right) that contains 100.0 g of water. The iron sample starts off at container 93.3 °C and the temperature of the water starts off at 22.0 °C. When the temperature of the water stops changing it's 25.7 °C. The pressure remains constant at 1 atm.
Required:
Calculate the mass of the iron sample. Be sure your answer is rounded to 2 significant digits.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, madisonrosamond99
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 14:30, Tooey2331
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 20:00, teacherpreacher
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
Do you know the correct answer?
A sample of iron, which has a specific heat capacity of 0.449 Jg^-1℃^-1, is put into a calorimeter (...
Questions in other subjects:

Mathematics, 26.07.2019 13:30





Mathematics, 26.07.2019 13:30

Mathematics, 26.07.2019 13:30



Chemistry, 26.07.2019 13:30