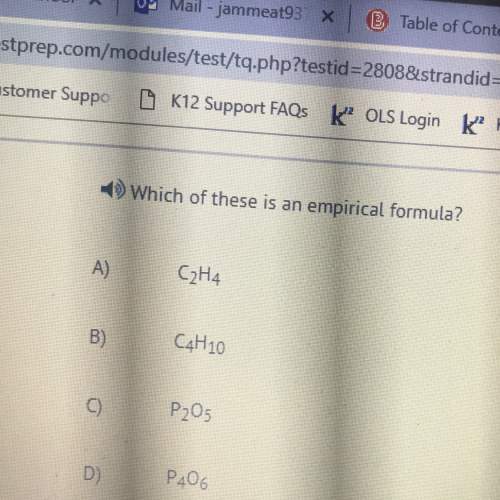
Chemistry, 05.02.2021 07:40, mauricestepenson791
Write the empirical formula for at least four ionic compounds that could be formed from the following ions: Pb4+, Fe2+, 103, ClO3

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, AbhiramAkella
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 22.06.2019 22:30, kristen17diaz
How many valence electrons are in atom of radon?
Answers: 1
Do you know the correct answer?
Write the empirical formula for at least four ionic compounds that could be formed from the followin...
Questions in other subjects:

Biology, 23.09.2020 01:01

Biology, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01

Computers and Technology, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01

History, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01

Social Studies, 23.09.2020 01:01